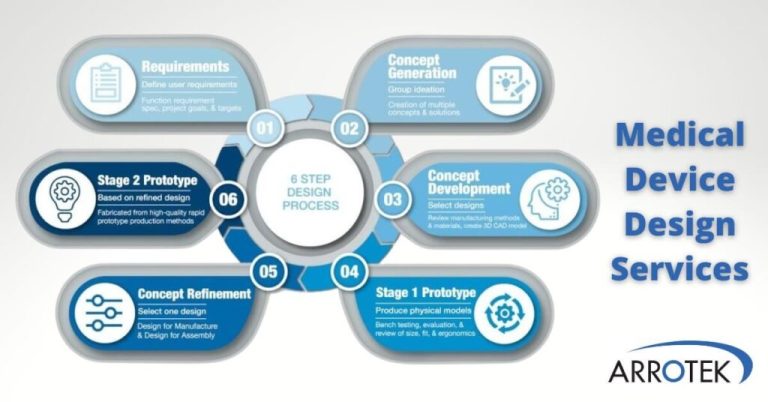
Medical Device Design Validation Explained
Medical device design validation is an essential part of the product development process. Effective and efficient design validation can help

Medical device design validation is an essential part of the product development process. Effective and efficient design validation can help